Water is essential in every industry. But in pharmaceutical manufacturing, biotechnology, medical device production, and laboratory research – water is not just a utility. It is a raw material. And like every raw material used in the production of products that go into human bodies, it must meet a precise, rigorously defined quality standard.
That standard is USP Grade Water.
In everyday applications – drinking, cooking, industrial cooling, or general commercial use – water quality is measured against relatively broad parameters. Safe TDS levels for drinking water, acceptable hardness for industrial use, basic bacterial safety for public consumption. These are important standards, but they are broad ones. A small deviation above an acceptable limit might result in slightly harder water or slightly less pleasant taste – not a crisis.
In pharmaceutical manufacturing, the tolerance for deviation is essentially zero. A trace ionic contaminant that would be entirely irrelevant in a glass of drinking water can compromise the stability of an active pharmaceutical ingredient. A bacterial endotoxin that would cause no harm if consumed can trigger a life-threatening fever response when introduced intravenously through a contaminated injectable product. The water used in pharmaceutical production must therefore meet specifications so precise and so strict that achieving and maintaining them requires purpose-engineered, multi-stage water treatment systems – not standard RO purifiers.
At the heart of every system designed to produce USP Grade Water reliably and consistently is a Reverse Osmosis (RO) system. But as we will explain in detail, RO alone is not sufficient – and understanding why, and what additional technologies are required, is essential for any pharmaceutical manufacturer, hospital, laboratory, or industrial facility that needs to produce or procure USP-grade water.
This blog by Bangalore Aqua – Karnataka’s No. 1 water treatment company – breaks down everything you need to know about USP Grade Water, the TDS control challenge at its core, and the complete purification process that makes USP compliance achievable in real-world industrial and pharmaceutical environments.
What Is USP Grade Water?
USP stands for United States Pharmacopeia – the organisation that establishes legally enforceable quality standards for pharmaceutical ingredients, excipients, drug products, and the water used in their manufacture. While the USP is an American organisation, its standards are globally recognised and adopted – and in India, they are referenced by pharmaceutical manufacturers operating under Schedule M GMP requirements, WHO GMP certification, and USFDA approval.
In simple terms, USP Grade Water is water whose chemical, physical, and biological properties meet the pharmacopeial purity specifications required for use in pharmaceutical manufacturing, medical device production, laboratory testing, and clinical applications.
It is not simply “very clean” water. It is water with defined, measured, and continuously monitored purity parameters – produced by a validated purification process, stored in validated equipment, and distributed through a validated system designed to prevent recontamination.
The United States Pharmacopeia defines several grades of pharmaceutical water. The two most widely relevant for Indian pharmaceutical and healthcare industries are Purified Water (PW) and Water for Injection (WFI) – and understanding the difference between them is the starting point for understanding USP water system design.
What Is TDS – And Why Does It Matter for USP Water?
TDS – Total Dissolved Solids – refers to the total concentration of all dissolved substances in water. This includes inorganic salts such as calcium, magnesium, sodium, potassium, chloride, sulfate, and bicarbonate, as well as dissolved organic compounds, heavy metals, and any other dissolved material present in the water.
TDS is measured in milligrams per litre (mg/L) or equivalently in parts per million (ppm). In the context of USP water, TDS is most critically measured and monitored through electrical conductivity – expressed in microsiemens per centimetre (μS/cm) – or its inverse, resistivity, expressed in megaohm-centimetres (MΩ·cm). Because dissolved ionic substances conduct electricity, conductivity is a real-time, continuous, and highly sensitive proxy for dissolved ion concentration – making it the primary quality monitoring parameter in pharmaceutical water systems.
To understand why TDS control is so critical in USP water applications, consider what dissolved solids actually do in a pharmaceutical context:
Ionic Interference with Drug Formulation – Dissolved ions from inorganic salts can interact with active pharmaceutical ingredients – altering pH, affecting solubility, accelerating degradation, and compromising the stability and efficacy of the final drug product. Even trace ionic concentrations that are perfectly safe for drinking can be chemically significant in pharmaceutical formulation.
Organic Contamination Risks – Dissolved organic compounds – from water treatment chemicals, naturally occurring organic matter, or industrial contamination – can react with pharmaceutical ingredients, affect product stability, contribute to discoloration, and cause formulations to fail quality testing.
Microbial Growth Substrate – Dissolved organic nutrients in water provide the substrate that supports bacterial growth and biofilm formation in storage tanks and distribution pipework. Water with higher dissolved organic content is significantly more prone to microbial contamination – a critical compliance risk in pharmaceutical water systems.
Analytical Interference – Water used as a solvent or diluent in laboratory testing and analytical chemistry must be free of ionic and organic interference to ensure accurate, reproducible results. Even sub-ppm levels of dissolved contaminants can affect analytical precision.
USP Grade Water Standards – Purified Water & WFI
USP Purified Water (PW)
Purified Water is the standard grade of USP water used in the manufacture of non-sterile pharmaceutical products, for equipment cleaning and rinsing, in compounding pharmacies, and in laboratory and analytical applications where high-purity water is required but injectable-grade sterility and endotoxin control are not.
The key USP specifications for Purified Water are:
| Parameter | USP Purified Water Limit |
| Conductivity | ≤ 1.3 μS/cm at 25°C |
| Total Organic Carbon (TOC) | ≤ 500 ppb (0.5 mg/L) |
| Nitrates | ≤ 0.2 ppm |
| Heavy Metals | ≤ 0.1 ppm |
| Microbial Action Level | ≤ 100 CFU/mL |
| Endotoxins | Not specified |
Purified Water must be produced by a validated process – most commonly reverse osmosis combined with electrodeionisation, ion exchange, or distillation – and must be stored and distributed in a system designed to prevent microbial and chemical recontamination.
USP Water for Injection (WFI)
Water for Injection is the highest purity grade of USP water – required for the manufacture of sterile injectable pharmaceutical products, ophthalmic preparations, and other applications where product contact with the water carries a direct patient safety risk. WFI meets all the same chemical purity specifications as Purified Water but with two critically additional requirements – a much stricter microbial limit and a mandatory bacterial endotoxin limit.
| Parameter | USP WFI Limit |
| Conductivity | ≤ 1.3 μS/cm at 25°C |
| Total Organic Carbon (TOC) | ≤ 500 ppb (0.5 mg/L) |
| Bacterial Endotoxins | ≤ 0.25 EU/mL |
| Microbial Action Level | ≤ 10 CFU/100 mL |
| Heavy Metals | ≤ 0.1 ppm |
| Nitrates | ≤ 0.2 ppm |
The bacterial endotoxin limit is the defining additional requirement for WFI. Endotoxins are pyogenic substances derived from the cell walls of gram-negative bacteria. When introduced into the bloodstream through a contaminated injectable product, even extremely small quantities of endotoxin can trigger severe fever, hypotension, and septic shock responses. Standard RO and even EDI do not effectively remove endotoxins – which is why WFI production requires additional treatment steps including ultrafiltration or distillation beyond the standard RO + EDI train used for Purified Water.
Indian Pharmacopoeia & WHO GMP Alignment
In India, the Indian Pharmacopoeia (IP) defines Purified Water and Water for Injection specifications that are closely harmonised with USP standards. Indian pharmaceutical manufacturers operating under Schedule M GMP and those seeking USFDA, EU GMP, or WHO GMP certification must produce pharmaceutical water that meets these internationally harmonised specifications – making USP compliance both a domestic regulatory requirement and a global market access requirement for Indian pharma manufacturers.
Required TDS Levels for USP Grade Water

Understanding the TDS and conductivity levels required for USP compliance – and how dramatically different they are from typical source water in Bangalore and Karnataka – makes clear why a standard RO system alone is insufficient and why a properly engineered multi-stage purification system is the only viable approach.
| Water Source / Type | Typical Conductivity | Approximate TDS | USP Compliant? |
| Borewell Water (Bangalore/Karnataka) | 800 – 3000 μS/cm | 500 – 2000 ppm | No |
| Municipal Tap Water (Bangalore) | 200 – 600 μS/cm | 130 – 400 ppm | No |
| Single-Pass RO Permeate | 10 – 50 μS/cm | 5 – 30 ppm | No |
| Double-Pass RO Permeate | 2 – 10 μS/cm | 1 – 6 ppm | Borderline |
| RO + EDI Output | 0.05 – 0.5 μS/cm | < 0.5 ppm | Yes – PW Grade |
| USP Purified Water Limit | ≤ 1.3 μS/cm | < 1 ppm | Required |
| USP WFI Limit | ≤ 1.3 μS/cm + endotoxin ≤ 0.25 EU/mL | < 1 ppm | Required |
The critical insight here is that even a well-operated single-pass RO system producing permeate at 10–50 μS/cm is still 10 to 40 times above the USP Purified Water conductivity limit of 1.3 μS/cm. Standard RO dramatically reduces TDS – but it does not get close to USP compliance on its own. Achieving USP-compliant conductivity requires RO combined with further polishing technology – most commonly Electrodeionisation (EDI) – to reliably bring conductivity below 1.3 μS/cm and maintain it there continuously.
The Role of the RO System in USP Water Production
Despite being insufficient alone, the RO system is the single most critical component in the USP water production train – and the quality of its design, operation, and maintenance determines the performance of every downstream stage.
Primary TDS Reduction – Doing the Heavy Lifting
The RO system performs the primary, bulk reduction of dissolved solids – taking source water from 200–3000 μS/cm conductivity down to 10–50 μS/cm in the permeate. This 95–99% reduction of the total ionic load is the most energy-intensive and technically demanding step in the purification process – and doing it correctly in the RO stage protects the downstream EDI unit from overload, extends its operational life, and ensures the complete system can maintain USP compliance economically and reliably.
Biological Pre-Treatment
RO membranes with molecular weight cut-offs in the 100–200 Dalton range effectively reject bacteria, viruses, colloidal particles, and larger organic molecules – providing the primary biological quality reduction that UV and ultrafiltration stages downstream then further address. Without this biological reduction at the RO stage, downstream disinfection systems would face a far greater bioburden and the risk of system contamination would increase substantially.
Organic Compound Reduction
RO membranes reject a significant proportion of dissolved organic compounds – making a meaningful contribution to the TOC reduction required to meet the USP limit of 500 ppb. In most pharmaceutical water applications, the RO stage reduces TOC to levels where downstream UV treatment can complete the reduction to compliance.
Protecting Downstream Equipment
By removing the bulk of dissolved ions, bacteria, and particulates, the RO system protects the downstream EDI, UV, and ultrafiltration components from the fouling, scaling, and biological challenges that would significantly shorten their operational life if they had to handle raw or minimally pre-treated water.
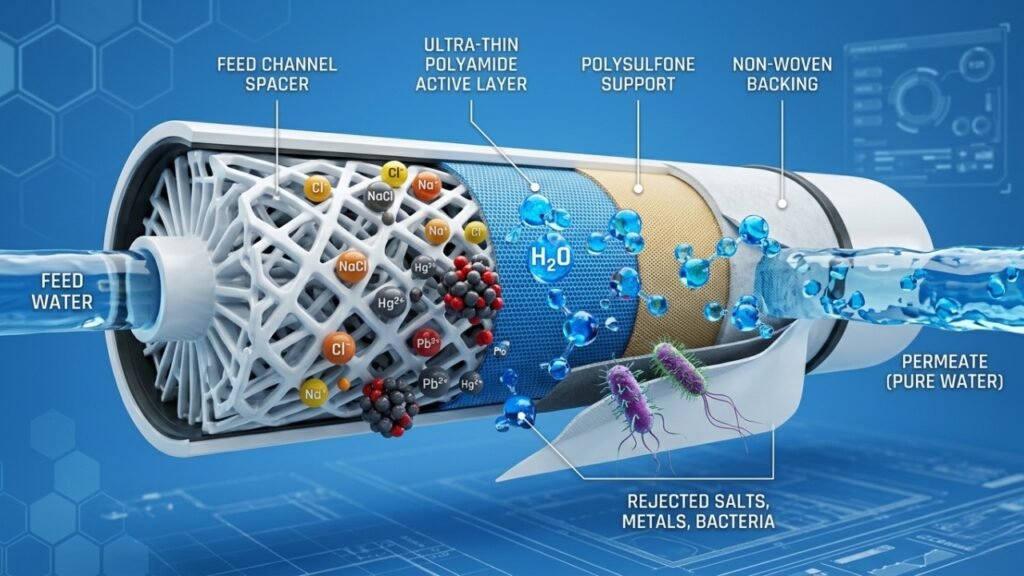
The Complete Process – RO + EDI + UV for USP Grade Water

Producing USP Purified Water that consistently meets pharmacopeial specifications in a real-world pharmaceutical or industrial environment requires a carefully designed, multi-stage purification train. Here is each stage explained in full:
Stage 1 – Pre-Treatment System
Multimedia Sand Filter – Removes suspended solids, sediment, and turbidity from raw source water that would foul downstream membranes and components prematurely.
Activated Carbon Filter – Removes chlorine, chloramines, and dissolved organic compounds. Chlorine removal is absolutely critical at this stage – chlorine irreversibly damages RO membranes, and its presence in the feed water even at low concentrations degrades membrane performance and lifespan over time.
Water Softener – Removes calcium and magnesium hardness from feed water. Hard water causes carbonate scaling on RO membranes – reducing performance and shortening membrane life. Softening the feed water upstream of the RO stage is standard practice for pharmaceutical water systems operating on Bangalore’s hard borewell or municipal water sources.
Antiscalant Dosing System – Controlled chemical dosing prevents residual scale formation from calcium carbonate, barium sulfate, silica, and other scale-forming compounds on the RO membrane surfaces.
Micron Cartridge Filter – A 5-micron or 1-micron cartridge filter immediately before the RO high-pressure pump removes any remaining particulates – protecting the pump and providing a final barrier before the membranes.
Stage 2 – Reverse Osmosis
The pre-treated feed water is pressurised by a high-pressure pump and forced through semi-permeable RO membranes – achieving 95–99% rejection of dissolved ions, organic molecules, bacteria, and viruses.
In pharmaceutical-grade RO systems, several design features beyond standard industrial RO are standard practice:
Double-Pass RO Configuration – Two RO stages in series, where the permeate of the first pass becomes the feed to the second pass. Double-pass RO achieves permeate conductivity of 1–5 μS/cm – approaching USP limits and dramatically reducing the polishing load on the downstream EDI unit.
Continuous Recirculation Loop – A portion of the product water continuously recirculates through the RO loop to prevent stagnation and microbial growth in the system during low-demand periods.
Online Conductivity Monitoring with Auto-Divert – Continuous real-time monitoring of permeate conductivity with automatic diversion of any off-spec water to drain – ensuring that only water meeting quality criteria proceeds to downstream polishing stages.
Sanitary Design – Pharmaceutical-grade RO units use sanitary fittings, food-grade materials, and design features that facilitate cleaning and sanitisation – minimising biofilm formation and supporting GMP compliance.
Stage 3 – Electrodeionisation (EDI)
EDI is the technology that bridges the gap between standard RO output and USP Purified Water compliance. It is a continuous, chemical-free ion removal process that combines ion exchange resin, ion-selective membranes, and a direct electrical current to remove the residual ionic contaminants from the RO permeate that the membrane alone cannot eliminate.
How EDI Works
An EDI module contains alternating diluting and concentrating compartments separated by cation and anion exchange membranes. Mixed ion exchange resin in the diluting compartments captures residual ions from the RO permeate as it flows through. A direct electrical current applied across the module continuously regenerates the resin electrically – driving the captured ions through the ion-selective membranes into the adjacent concentrating compartments, from which they are continuously flushed to drain as a small waste stream.
The result is a continuous, chemical-free polishing process that reliably achieves product water conductivity of 0.05–0.1 μS/cm – well within the USP Purified Water requirement of ≤ 1.3 μS/cm – without the batch regeneration cycles, chemical handling, and operational complexity of conventional mixed-bed deionisation.
Key Advantages of EDI Over Conventional Mixed-Bed Deionisation
No chemical regeneration required – eliminates hazardous acid and caustic handling, reducing operating costs, safety risks, and regulatory compliance burden significantly
Continuous, uninterrupted operation – no offline regeneration downtime means continuous, consistent product water supply for manufacturing operations
More consistent output quality – EDI delivers stable, consistent resistivity without the quality variation between regeneration cycles that conventional deionisation exhibits
Lower TOC contribution – EDI does not introduce TOC from resin degradation the way aged conventional mixed-bed resins do, maintaining TOC compliance more reliably over time
Lower long-term operating cost – despite higher capital cost compared to mixed-bed deionisation, the elimination of chemical procurement, waste disposal, and regeneration labour makes EDI more economical over a 5–10 year operational horizon
Stage 4 – Ultraviolet (UV) Disinfection
UV disinfection systems are installed at multiple critical points in the USP water production and distribution system – delivering two distinct but equally important functions depending on the wavelength used:
UV at 254 nm – Microbial Inactivation – UV light at 254 nm is germicidal – it disrupts the DNA of bacteria, viruses, and other microorganisms, preventing them from reproducing and effectively eliminating them from the water stream. UV units are installed after RO, after EDI, and at strategic points in the distribution loop to maintain continuous microbial control throughout the system.
UV at 185 nm – TOC Reduction – UV light at 185 nm photooxidises dissolved organic compounds in the water – breaking them down into carbon dioxide and water and progressively reducing Total Organic Carbon levels toward and below the USP limit of 500 ppb. For systems where the combined RO and EDI treatment does not fully achieve TOC compliance, a 185 nm UV unit provides the additional organic reduction required.
Stage 5 – Ultrafiltration (UF) for WFI Grade
For applications requiring Water for Injection purity – where bacterial endotoxin removal to ≤ 0.25 EU/mL is a mandatory specification – ultrafiltration using membranes with a molecular weight cut-off of approximately 6,000–10,000 Daltons is employed as the final polishing step.
UF membranes effectively reject bacterial endotoxins – which are large lipopolysaccharide molecules typically in the 10,000–1,000,000 Dalton molecular weight range – that pass through RO membranes completely. The USP has recognised membrane-based WFI production using RO + EDI + UF as an equivalent alternative to the traditional multi-effect distillation method – allowing pharmaceutical manufacturers to produce WFI-grade water without the high capital and operating costs associated with distillation systems.
Stage 6 – Storage & Distribution System
Producing USP-grade water is only half the challenge. Maintaining its purity through storage and distribution to every point of use in the facility is equally critical – and is where many pharmaceutical water systems fail compliance.
Key design requirements for USP water storage and distribution include:
316L Stainless Steel Tanks and Pipework – Pharmaceutical-grade stainless steel that does not leach contaminants, is resistant to sanitisation chemicals, and can be passivated to minimise surface reactivity and microbial adhesion
Continuous Recirculation at Minimum 1–2 m/s – Distribution loops maintain constant water circulation at velocities that prevent biofilm formation – the single most common cause of microbial out-of-specification events in pharmaceutical water systems
Continuous UV in the Distribution Loop – Inline UV units in the recirculation loop provide continuous microbial control throughout the distribution system
Hot Water Sanitisation Capability – Systems designed to periodically circulate hot water at ≥ 80°C throughout the entire loop to eliminate established biofilm and maintain long-term microbial compliance
Online Continuous Monitoring – Real-time conductivity and TOC monitoring at critical sampling points with automated diversion of any out-of-specification water – providing continuous assurance of compliance between formal quality testing intervals
Why TDS Control Is Critical in USP Water Systems
TDS control – specifically conductivity management – is not just a quality specification to be tested periodically and filed in a report. It is the real-time, continuous indicator of your entire purification system’s performance – and the earliest warning signal that something is going wrong.
Here is why TDS control deserves to be treated as the operational heartbeat of your USP water system:
Conductivity Is Your System Health Monitor
Every component in your USP water purification train contributes to TDS reduction. When conductivity rises above its normal operating range at any monitoring point, it signals a specific problem at a specific stage – a fouled RO membrane, an exhausted EDI unit, a depleted softener resin bed, a failed activated carbon filter allowing chlorine breakthrough to the membranes. Continuous conductivity monitoring gives you real-time visibility into the health of every treatment stage – allowing you to identify and address problems before they result in batch failures or regulatory findings.
Conductivity Is the Foundation of USP Compliance
The USP conductivity limit of ≤ 1.3 μS/cm for Purified Water and WFI is not a guideline – it is a pharmacopeial specification. Product batches manufactured using water that exceeded this limit at the time of manufacture are non-compliant – regardless of how the finished product tests. In a regulatory inspection, out-of-specification conductivity data without documented corrective action is a serious GMP finding that can result in warning letters, consent decrees, or product recalls.
Conductivity Changes Predict Downstream Quality Problems
Conductivity is often the first parameter to go out of specification when system performance deteriorates – preceding microbial exceedances and TOC failures in many cases. A facility that monitors and responds to conductivity trends proactively can often prevent a conductivity trend from becoming a microbial or TOC compliance failure – avoiding the far more serious consequences of biological contamination.
TDS Control Protects Your Downstream Equipment
Maintaining tight TDS control at each stage of the purification train protects downstream equipment from overload. If RO performance deteriorates and conductivity of the permeate rises, the EDI unit faces a higher ionic load than it was designed for – leading to faster deterioration of its ion exchange resin and ion-selective membranes. Proactive TDS monitoring at the RO outlet allows you to detect membrane degradation early and schedule maintenance before the EDI unit is damaged.
TDS Control Directly Impacts Product Quality & Patient Safety
Ultimately, TDS control in a pharmaceutical water system is not an abstract engineering parameter – it is a patient safety measure. The water used to formulate injectable drugs, sterile solutions, and pharmaceutical products comes into direct contact with the human body. Every ionic, organic, or biological contaminant that passes through an inadequately controlled water system has the potential to end up in a patient. TDS control is the continuous, real-time guard against that outcome.
Common Mistakes in USP Water System Design & Operation
Even well-intentioned pharmaceutical facilities and industrial users make predictable mistakes in designing, operating, and maintaining USP water systems. Here are the most common ones – and why they matter:
Mistake 1 – Relying on Single-Pass RO Without Downstream Polishing
The most common and consequential mistake is assuming that a standard single-pass RO system is sufficient to produce USP Purified Water. As the conductivity comparison table earlier in this article clearly shows, single-pass RO permeate typically has conductivity of 10–50 μS/cm – far above the USP limit of 1.3 μS/cm. Facilities that deploy single-pass RO without EDI or equivalent polishing and claim USP Purified Water compliance are either testing infrequently and getting lucky, or not testing properly at all. This is a critical GMP compliance risk.
Mistake 2 – Inadequate Pre-Treatment Upstream of RO
Installing a high-quality RO system without adequate pre-treatment is one of the most expensive mistakes a facility can make. Chlorine in the feed water destroys RO membranes – typically within weeks of exposure. Hard water causes rapid carbonate scaling that permanently reduces membrane performance. Suspended solids foul membrane surfaces and reduce flux. A facility that skimps on pre-treatment to reduce upfront costs invariably spends far more on premature membrane replacement and system downtime than the pre-treatment equipment would have cost.
Mistake 3 – No Continuous Conductivity Monitoring
Some facilities monitor conductivity only during periodic quality testing – daily, weekly, or even less frequently. Between testing intervals, the system operates without any real-time quality assurance. A membrane failure, a regeneration failure, or an EDI performance drop that occurs between tests results in non-compliant water being used in manufacturing for days or weeks before the problem is detected. Continuous online conductivity monitoring with auto-divert capability is not optional in a GMP-compliant pharmaceutical water system – it is a fundamental design requirement.
Mistake 4 – Poor Storage & Distribution System Design
Many pharmaceutical facilities invest appropriately in the purification train but then store and distribute the purified water through a system that recontaminates it – carbon steel tanks that rust and leach iron, low-velocity distribution loops where biofilm establishes, dead legs in pipework where water stagnates, or storage tanks without proper vent filtration that allow airborne contamination. The purification system can produce perfectly compliant water that arrives at the point of use completely out of specification because the distribution system undoes the work of the purification train.
Mistake 5 – Neglecting System Sanitisation
USP water systems must be sanitised regularly – either by periodic hot water circulation, chemical sanitisation with approved agents, or ozonation. Facilities that commission a system, achieve compliance at startup, and then neglect ongoing sanitisation typically see microbial counts creep upward over months as biofilm establishes progressively in the distribution loop – eventually resulting in microbial out-of-specification events that are expensive and disruptive to remediate.
Mistake 6 – Skipping Validation
A USP water system in a GMP pharmaceutical environment must be validated – demonstrating through documented commissioning, qualification, and performance testing that it consistently produces water meeting USP specifications under normal operating conditions. Facilities that skip or inadequately document validation have no documented basis for their compliance claims – and face serious regulatory exposure in any inspection.
Mistake 7 – Ignoring Seasonal Feed Water Variation
In Bangalore and across Karnataka, Mysuru, Yadgir, Vijayanagara, Chamarajanagar, feed water quality varies seasonally – TDS, hardness, turbidity, and bacterial load all change with the monsoon and the dry season. A USP water system designed and commissioned for one set of feed water conditions may perform differently when feed water quality changes significantly. Facilities that do not monitor feed water quality regularly and adjust treatment parameters accordingly risk performance drift in their purification system during periods of challenging feed water quality.
Conclusion
USP Grade Water is not simply clean water. It is precisely defined, continuously monitored, and rigorously controlled water – produced through a validated multi-stage purification process designed to meet pharmacopeial specifications that protect the integrity of pharmaceutical products and the safety of the patients who receive them.
At the foundation of every effective USP water production system is a properly designed, correctly installed, and well-maintained Reverse Osmosis system – performing the primary bulk reduction of dissolved solids, biological contaminants, and organic compounds that makes downstream USP compliance achievable. But RO alone is not sufficient. Achieving the USP Purified Water conductivity limit of ≤ 1.3 μS/cm requires RO combined with EDI for ionic polishing, UV for biological control and TOC reduction, and – for WFI applications – ultrafiltration for endotoxin removal.
TDS control, expressed through continuous conductivity monitoring, is the operational heartbeat of every USP water system – the real-time indicator of system health, the earliest warning of performance deterioration, and ultimately a direct measure of patient safety protection.
For pharmaceutical manufacturers, hospitals, biotechnology facilities, and industrial users in Bangalore and across Karnataka who need to design, install, or upgrade a USP-grade water production system – Bangalore Aqua brings the technical expertise, the validated system design capability, and the long-term maintenance support to deliver a system that performs to specification from day one and continues to do so reliably for years.
Clean water is not a marketing claim. It is a technical achievement – and achieving it to USP standards requires the right system, the right components, and the right partner.
Bangalore Aqua is that partner.
📞 Call / WhatsApp: +91 76763 93939 | +91 97387 04753
📧 Email: info@bangaloreaqua.com
🌐 Consult Our Water Treatment Experts: bangaloreaqua.com
Karnataka’s No. 1 Water Treatment Company – Serving pharmaceutical manufacturers, hospitals, industries & communities across Bangalore and Karnataka since 2021.


